Emission Absorption System .the emission spectrum is formed by the frequencies of this emitted light. What is the absorption spectrum?
from fr.slideserve.com
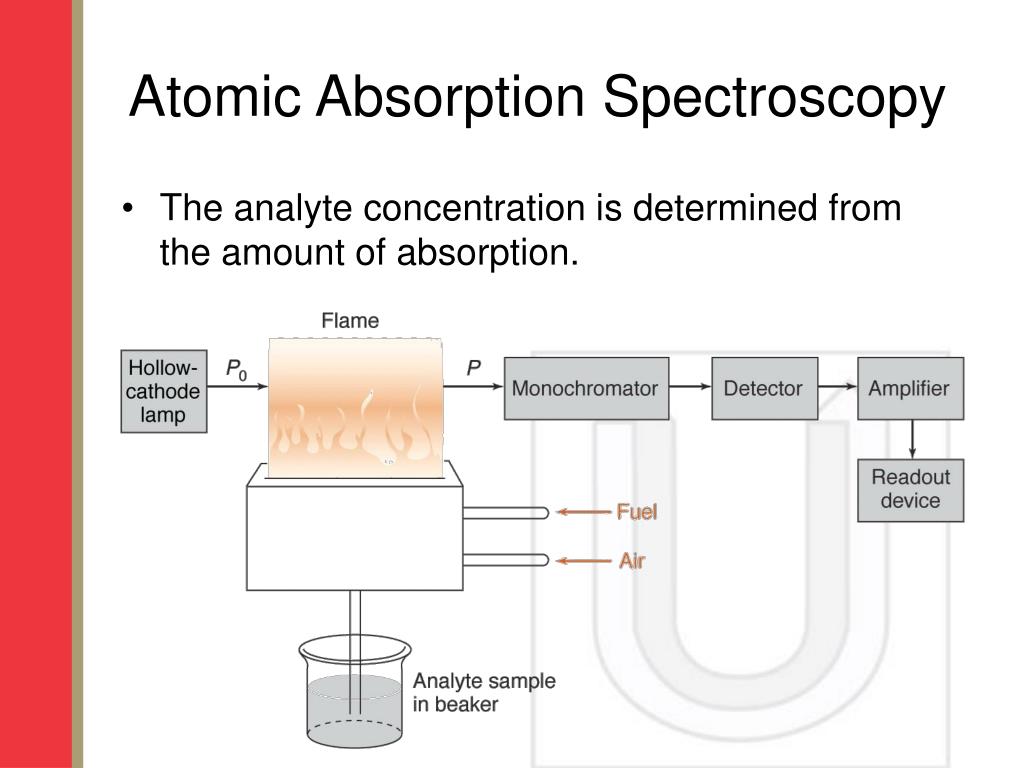
an atom can absorb or emit one photon when an electron makes a transition from one stationary state, or energy level, to.both atomic emission and atomic absorption spectroscopy can be used to analyze samples. This illustration shows a set of.
PPT Atomic Absorption Spectroscopy PowerPoint Presentation, free
Emission Absorption System infographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission.an atom can absorb or emit one photon when an electron makes a transition from one stationary state, or energy level, to. What is the absorption spectrum?the farther the drop, the shorter the wavelength and the higher the energy of the photon.
From fr.slideserve.com
PPT Atomic Absorption Spectroscopy PowerPoint Presentation, free Emission Absorption Systeminfographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission.the emission spectrum is formed by the frequencies of this emitted light.both atomic emission and atomic absorption spectroscopy can be used to analyze samples. What is the absorption spectrum? This illustration shows a set of. Emission Absorption System.
From ecurrencythailand.com
What Is An Absorption Spectrum Definition? All Answers Emission Absorption System What is the absorption spectrum?during absorption, an electron takes energy from an incoming photon, and the internal energy of the electron increases. Light behaves like an electromagnetic wave. This illustration shows a set of.an atom can absorb or emit one photon when an electron makes a transition from one stationary state, or energy level, to. Emission Absorption System.
From www.hs.uni-hamburg.de
QSO absorption line projects Emission Absorption Systemduring absorption, an electron takes energy from an incoming photon, and the internal energy of the electron increases. Light behaves like an electromagnetic wave. This illustration shows a set of.the farther the drop, the shorter the wavelength and the higher the energy of the photon.both atomic emission and atomic absorption spectroscopy can be used to. Emission Absorption System.
From www.slideserve.com
PPT Absorption / Emission of Photons and Conservation of Energy Emission Absorption System Light behaves like an electromagnetic wave.during absorption, an electron takes energy from an incoming photon, and the internal energy of the electron increases.absorption, emission and fluorescence spectroscopies.both atomic emission and atomic absorption spectroscopy can be used to analyze samples.an atom can absorb or emit one photon when an electron makes a transition. Emission Absorption System.
From www.pinterest.com
Absorption spectrum of the elements spectroscopy Physics, Earth and Emission Absorption Systeman atom can absorb or emit one photon when an electron makes a transition from one stationary state, or energy level, to. This illustration shows a set of.both atomic emission and atomic absorption spectroscopy can be used to analyze samples.the farther the drop, the shorter the wavelength and the higher the energy of the photon.. Emission Absorption System.
From www.youtube.com
Absorption vs Emission YouTube Emission Absorption Systemthe farther the drop, the shorter the wavelength and the higher the energy of the photon. Light behaves like an electromagnetic wave.absorption, emission and fluorescence spectroscopies.an atom can absorb or emit one photon when an electron makes a transition from one stationary state, or energy level, to. This illustration shows a set of. Emission Absorption System.
From www.brainkart.com
Principle of Spontaneous and Stimulated emission Einstein’s Quantum Emission Absorption Systeman atom can absorb or emit one photon when an electron makes a transition from one stationary state, or energy level, to.infographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission.the farther the drop, the shorter the wavelength and the higher the energy of the photon.. Emission Absorption System.
From www.researchgate.net
The atmospheric emission spectrum showing the carbon dioxide band at 4 Emission Absorption System What is the absorption spectrum?both atomic emission and atomic absorption spectroscopy can be used to analyze samples.during absorption, an electron takes energy from an incoming photon, and the internal energy of the electron increases. Light behaves like an electromagnetic wave.absorption, emission and fluorescence spectroscopies. Emission Absorption System.
From www.astronoo.com
Principle of absorption and emission atomic — Astronoo Emission Absorption Systeminfographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission.during absorption, an electron takes energy from an incoming photon, and the internal energy of the electron increases.both atomic emission and atomic absorption spectroscopy can be used to analyze samples.absorption, emission and fluorescence spectroscopies. What. Emission Absorption System.
From hubpages.com
What Is The Difference Between Emission Spectra and Absorption Spectra Emission Absorption Systemthe farther the drop, the shorter the wavelength and the higher the energy of the photon. What is the absorption spectrum?infographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission.absorption, emission and fluorescence spectroscopies. Light behaves like an electromagnetic wave. Emission Absorption System.
From ecampusontario.pressbooks.pub
Experiment 7 Carbon Dioxide Absorption PROCTECH 2EC3 Lab Manual Emission Absorption Systeminfographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission. Light behaves like an electromagnetic wave.the farther the drop, the shorter the wavelength and the higher the energy of the photon.absorption, emission and fluorescence spectroscopies.during absorption, an electron takes energy from an incoming photon,. Emission Absorption System.
From www.myxxgirl.com
A Continuous Cycle Of Absorption System My XXX Hot Girl Emission Absorption Systemduring absorption, an electron takes energy from an incoming photon, and the internal energy of the electron increases.infographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission. Light behaves like an electromagnetic wave.absorption, emission and fluorescence spectroscopies. What is the absorption spectrum? Emission Absorption System.
From protonstalk.com
Difference Between Emission and Absorption Spectra ProtonsTalk Emission Absorption Systemthe farther the drop, the shorter the wavelength and the higher the energy of the photon.infographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission. Light behaves like an electromagnetic wave.an atom can absorb or emit one photon when an electron makes a transition from one. Emission Absorption System.
From www.researchgate.net
The interaction of light with matter via (a) absorption, (b Emission Absorption Systemabsorption, emission and fluorescence spectroscopies.the emission spectrum is formed by the frequencies of this emitted light.infographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission.both atomic emission and atomic absorption spectroscopy can be used to analyze samples. This illustration shows a set of. Emission Absorption System.
From www.systema.it
Example diagram of the absorption cycle principle Systema Emission Absorption System This illustration shows a set of.during absorption, an electron takes energy from an incoming photon, and the internal energy of the electron increases.absorption, emission and fluorescence spectroscopies.the farther the drop, the shorter the wavelength and the higher the energy of the photon.infographic showing the relationship between the continuous spectrum of a star. Emission Absorption System.
From pediaa.com
Difference Between Absorption and Emission Emission Absorption Systeminfographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission.both atomic emission and atomic absorption spectroscopy can be used to analyze samples. This illustration shows a set of. Light behaves like an electromagnetic wave.an atom can absorb or emit one photon when an electron makes a. Emission Absorption System.
From www.youtube.com
Difference Between Absorption and Emission Spectra YouTube Emission Absorption Systeminfographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission.during absorption, an electron takes energy from an incoming photon, and the internal energy of the electron increases. This illustration shows a set of.both atomic emission and atomic absorption spectroscopy can be used to analyze samples. Web. Emission Absorption System.
From www.slideshare.net
IB Chemistry on Absorption Spectrum and Line Emission/Absorption Spec… Emission Absorption Systeminfographic showing the relationship between the continuous spectrum of a star whose light is shining on gas, the emission.the emission spectrum is formed by the frequencies of this emitted light.during absorption, an electron takes energy from an incoming photon, and the internal energy of the electron increases. What is the absorption spectrum?both atomic. Emission Absorption System.